GlassBone™ Injectable Putty
Injectable Putty for bone regeneration
Inspired by an innovative and reliable technology, GlassBone™ Injectable Putty is indicated in spine surgeries.
Composed of bioactive glass 45S5 and a polymer, it is 100% synthetic and completely assimilable by the body.
Bioactive Glass is made of silicium, calcium, sodium and phosphorus minerals, which are naturally presents in the human body and ensure excellent biocompatibility and osteoconduction.

GlassBone™ Injectable Putty is composed of 45S5 bioactive glass and a polymer. This composite technology allows it to be very easily malleable and applicable in complex defects. It is sold directly in the syringe, ready to use.
GlassBone™ Injectable Putty provides surgeons and patients with optimal safety during implantation for a natural and effective bone regeneration.
GlassBone™ Injectable Putty is developed and manufactured in France with local partners to ensure optimum quality.
GlassBone™, Bone graft substitute is a medical device class III, manufactured by NORAKER® and whose conformity assessment was conducted by GMED (0459).
Glassbone™ is indicated for filling bone defects. Read the instructions supplied with the product for complete information on indications, cons-indications, warnings and precautions, and adverse effects.
Bioactive Glass : the key technology to regenerate your bones
Intended use
GlassBone™ Injectable Putty is a synthetic, bioactive and absorbable bone substitute, intended for the filling, reconstruction and/or fusion of bone defects or gaps in the skeletal system, in spine surgery.
Indications and target population
Loss or lack of bone substance for bone defects of traumatic, pathological or surgical origin when autologous solutions are not applicable or sufficient neurosurgery in children and adult population:
- Fusion or reconstruction of deformities and degenerative diseases in spine
- Filling after surgical bone defect (donor sites after removal of autograft, trepanation, …)
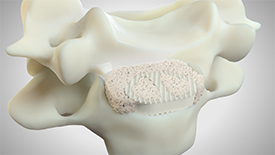
Bioactive Bone Substitute

GlassBone™ Injectable Putty is ready to use. It can be directly injected through the serynge.